Description
CJC-1295 (No DAC) – Short-Acting Growth Hormone Releasing
Description
CJC-1295 (No DAC) is a modified GHRH analog engineered to stimulate endogenous growth hormone release from the anterior pituitary. It binds to GHRH receptors on somatotroph cells, enhancing natural pulsatile GH secretion. The No DAC version lacks the drug affinity complex that prolongs half-life in the DAC formulation, resulting in shorter systemic activity. This shorter-acting profile allows researchers to study physiologic pulse amplification rather than sustained hormone elevation. CJC-1295 (No DAC) is often investigated in experimental models examining growth hormone rhythm optimization, IGF-1 pathway engagement, and endocrine recovery signaling.
Mechanism of Action
CJC-1295 (No DAC) stimulates growth hormone release by binding to GHRH receptors in the pituitary gland. This activation enhances natural GH pulses without directly supplying exogenous hormone.
Benefits
Enhances natural pulsatile growth hormone secretion
CJC-1295 (No DAC) amplifies endogenous growth hormone release by directly stimulating the GHRH receptor pathway in the anterior pituitary. Growth hormone is physiologically secreted in discrete pulses, with peak amplitude occurring during deep sleep phases. Rather than producing continuous elevation, CJC-1295 increases the magnitude of these pulses while preserving their rhythmic nature. This pulsatile enhancement is central to maintaining receptor sensitivity and preventing desensitization associated with sustained hormone exposure. The short-acting profile allows GH levels to rise and fall within natural regulatory parameters.
Selective engagement of the GHRH receptor cascade
CJC-1295 (No DAC) binds specifically to the growth hormone–releasing hormone receptor (GHRH-R), a G-protein–coupled receptor expressed on somatotroph cells. Activation of this receptor increases adenylate cyclase activity, elevating intracellular cAMP levels. This rise in cAMP activates protein kinase A, leading to enhanced transcription and exocytosis of growth hormone. Unlike ghrelin-based peptides that act through the GHS-R1a receptor, CJC-1295 does not directly stimulate appetite-related hypothalamic pathways. This receptor specificity differentiates it mechanistically from GHRP compounds.
Preserves hypothalamic somatostatin regulation
The hypothalamus tightly controls GH release through a balance between GHRH stimulation and somatostatin inhibition. Because CJC-1295 stimulates endogenous pathways rather than bypassing them, somatostatin feedback remains active. This allows the body to modulate hormone output dynamically based on physiologic demand. Maintaining this inhibitory counterbalance is important in experimental models examining long-term endocrine stability.
Supports downstream IGF-1–mediated anabolic signaling
Once growth hormone is released, it stimulates hepatic production of insulin-like growth factor 1 (IGF-1). IGF-1 activates intracellular pathways including PI3K/Akt and mTOR, which regulate protein synthesis and cellular growth. Through this two-step endocrine cascade, CJC-1295 indirectly influences tissue adaptation and recovery-related signaling. This layered activation preserves physiological hierarchy rather than directly activating peripheral receptors.
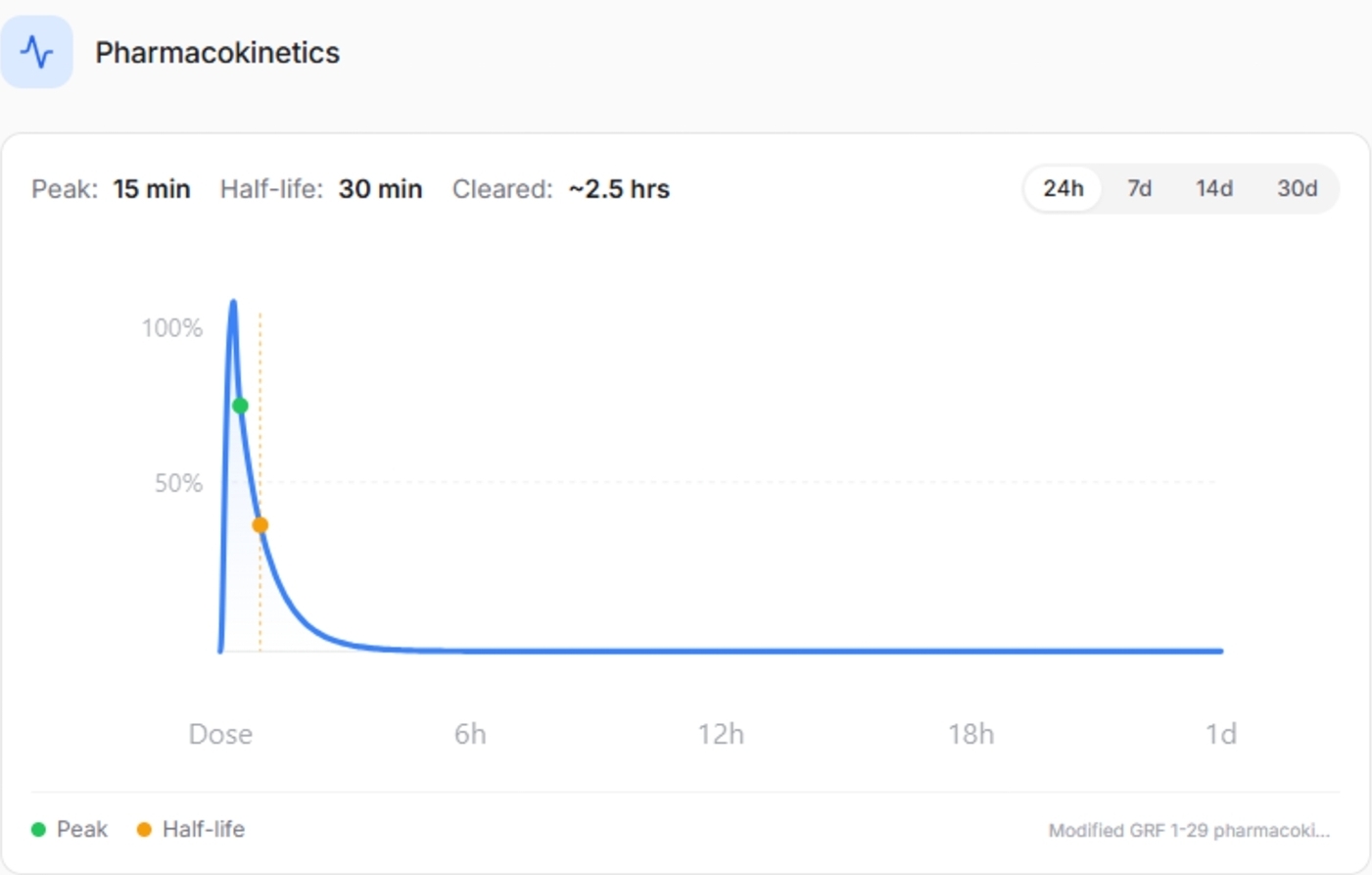
Short-acting profile enables pulse timing studies
Unlike the DAC-modified formulation, the No DAC version lacks albumin-binding extension and therefore has a shorter half-life. This transient activity allows researchers to evaluate timing-dependent endocrine responses and synchronization with circadian cycles. Pulse timing plays a critical role in how tissues respond to growth hormone exposure. By aligning with physiologic rhythms, CJC-1295 (No DAC) supports research focused on hormone pattern optimization rather than continuous stimulation.
Influences sleep-associated growth hormone peaks
Growth hormone secretion is closely linked to slow-wave sleep. Amplifying physiologic GH pulses may enhance the magnitude of nocturnal hormone peaks in experimental models. This connection places CJC-1295 within integrated sleep–endocrine research frameworks. Rather than inducing sleep directly, it operates within hormonal cycles that coincide with restorative phases.
Distinct from ghrelin mimetics and appetite-driven peptides
GHRP compounds stimulate growth hormone release via ghrelin receptor activation and may influence hunger pathways. CJC-1295 operates exclusively through the GHRH receptor pathway, avoiding direct activation of neuropeptide Y–mediated appetite signaling. This distinction makes it more focused on pituitary modulation than hypothalamic hunger circuits. The absence of strong appetite stimulation differentiates its systemic profile.
Facilitates structured endocrine modulation research
Because it enhances natural hormone pulses rather than replacing GH, CJC-1295 supports investigation into physiologic endocrine adaptation. The preservation of feedback loops, receptor sensitivity, and pulse rhythm allows researchers to examine nuanced hormonal modulation. This precision positions the No DAC variant within rhythm-aligned growth hormone research rather than sustained supraphysiologic exposure models.
Integrates within broader neuroendocrine coordination networks
Growth hormone interacts with metabolic regulation, connective tissue remodeling, and circadian timing systems. By amplifying endogenous GH pulses, CJC-1295 participates in this integrated neuroendocrine network. Its activity reflects coordinated communication between hypothalamus, pituitary, and peripheral tissues. This systems-level positioning differentiates it from peptides that act directly at peripheral receptors without upstream regulatory integration.
Peptide Interactions (Stack Suggestions)
| Peptide | Interaction | Description |
|---|---|---|
| Ipamorelin | Synergistic | Excellent combination – Ipamorelin (GHRP) + CJC-1295 (GHRH) provides complementary GH release through different receptor pathways |
| GHRP-6 | Synergistic | Strong synergy when combined – GHRP-6 amplifies the GH pulse initiated by CJC-1295 |
| GHRP-2 | Synergistic | Potent combination for maximizing growth hormone release through dual pathway activation |
| Hexarelin | Monitor Combination | Can be combined but monitor for excessive GH elevation due to Hexarelin’s potency |
| CJC-1295 with DAC | Avoid Combination | Never combine different CJC-1295 variants – choose based on desired release pattern |
| Tesamorelin | Use Caution | Both bind to pituitary GHRH receptors. Combining saturates receptors without amplifying GH release, risking receptor desensitization and blunted pulsatility. Choose one based on your protocol goals. |
| MK-677 | Compatible | Different mechanisms – MK-677 provides continuous ghrelin stimulation while CJC-1295 offers pulsatile GHRH |
Dosing & Reconstitution Guide
| Parameter | Details (CJC 1295 No DAC 5mg) |
|---|---|
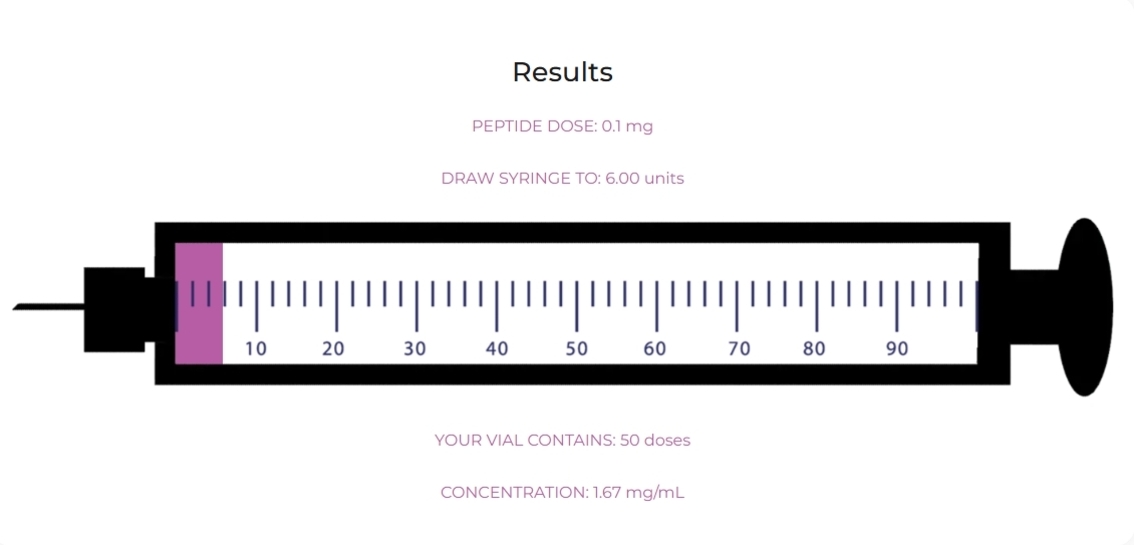
| Volume | Add 3.0 mL bacteriostatic water |
| Concentration | 1.67 mg/mL |
| Dose | 0.1 – 0.3 mg once daily |
| Cycle Length | 8 – 12 weeks |
Dosage & Protocols Variations
| Week | Daily Dose (mg) | Units (per injection) (mL) |
|---|---|---|
| Weeks 1–2 | 0.1 mg | 6 units (0.06 mL) |
| Weeks 3–4 | 0.15 mg | 9 units (0.09 mL) |
| Weeks 5–6 | 0.2 mg | 12 units (0.12 mL) |
| Weeks 7–12 | 0.25 – 0.30 mg | 15–18 units (0.15–0.18 mL) |
Suggested daily titration approach:
Start: 0.1–0.15 mg daily; increase by ~0.05 mg every 1–2 weeks as tolerated.
Target: 0.2–0.3 mg daily by Weeks 5–12.
Frequency: Once per day (subcutaneous), preferably at bedtime.
Cycle Length: 8–12 weeks; optional extension to 16 weeks.
Timing: Administer at bedtime to align with natural nocturnal GH pulses; rotate injection sites.
Storage Instructions
Proper storage preserves peptide quality and stability.
- Lyophilized: Store at −20 °C in dry, dark conditions; minimize moisture exposure.
- Reconstituted: Refrigerate at 2–8 °C; use within 4–6 weeks; avoid freeze–thaw.
- Allow vials to reach room temperature before opening to reduce condensation uptake.